 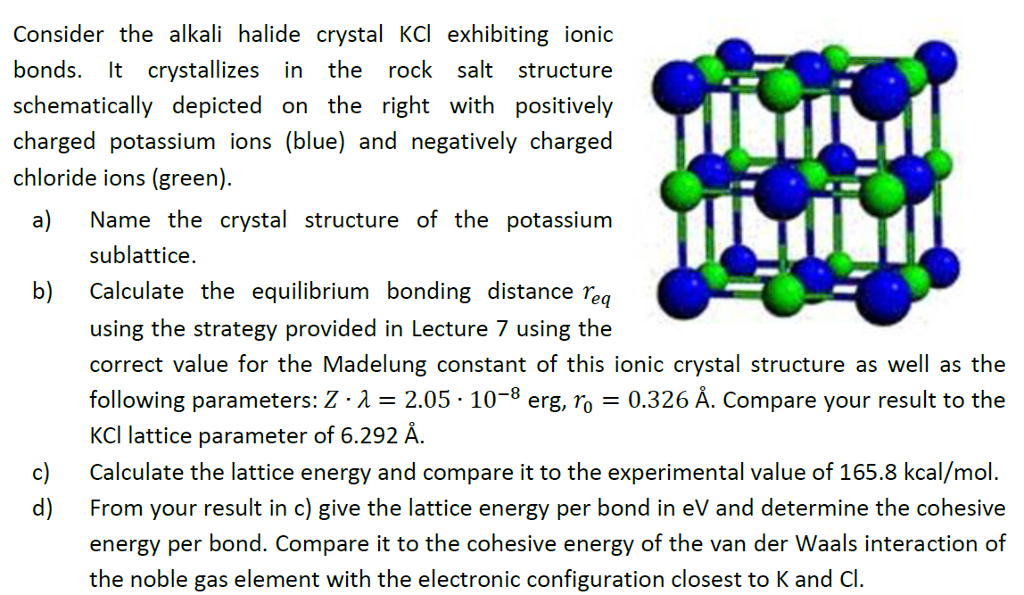 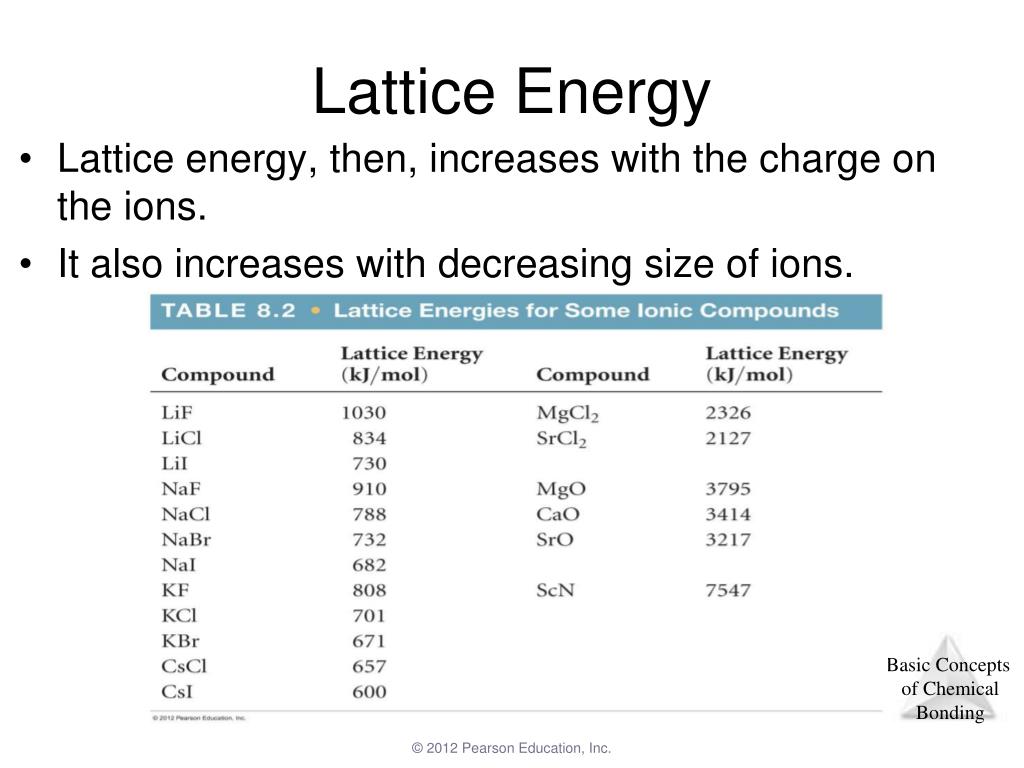
higher ionization of lithium is more than compensated by the higher lattice energy and pushing the equilibrium as potassium vapor partial pressure is being decreased by vapor condensation. Thermodynamic parameters are shown in the table below. The lattice energies (Delta Ho lattice) for sodium chloride and potassium chloride are -788 kJ and -717 kJ, respectively. With potassium vapors being eliminated by condensation on colder surfaces of the apparatus, used in the video. What is the lattice energy for NaCl The lattice energy of sodium chloride is 789 kJ/mol. That is because higher ionization energy is more than compensated by more negative hydration enthalpy of $\ce$$ The lattice energy for KCl is 715 kJ mol-1. But lithium has more negative standard redox potential (which applies in water context only). In order to continue to use our website, we ask you to confirm your identity as a person. So the lattice energy of KCl is +838.5 kJ/mol. Below is a diagram of the Born-Haber cycle for the formation of KCl: Now calculate the lattice by adding the total energy value and subtracting the formation heat. Potassium reacts more violently with water than lithium. Some energy (lattice energy) will also be released when KCl forms from potassium and chloride ions. Consequently, the lattice strength of CaS would be. As Ca, S have 2 units of charge while K, Cl have one and there is not much of a difference in size ( atleast not enough to overcome the doubling of the charge), the charge density of Ca and S ions would be more. TNT is much more reactive than coal, but released specific energy during coal burning is 10 times bigger than during TNT explosion. The Lattice Energy of Ionic Compounds is directly proportion to the Charge Density. In the discussion of lattice energy, we consider the ions separated into a gas form whereas in the dissolution process, the ions are also separated, but this time into ions dispersed in a medium with solvent molecules between ions. It is odorless and has a white or colorless vitreous crystal appearance. Relating Hydration Energy to Lattice Enthalpy. 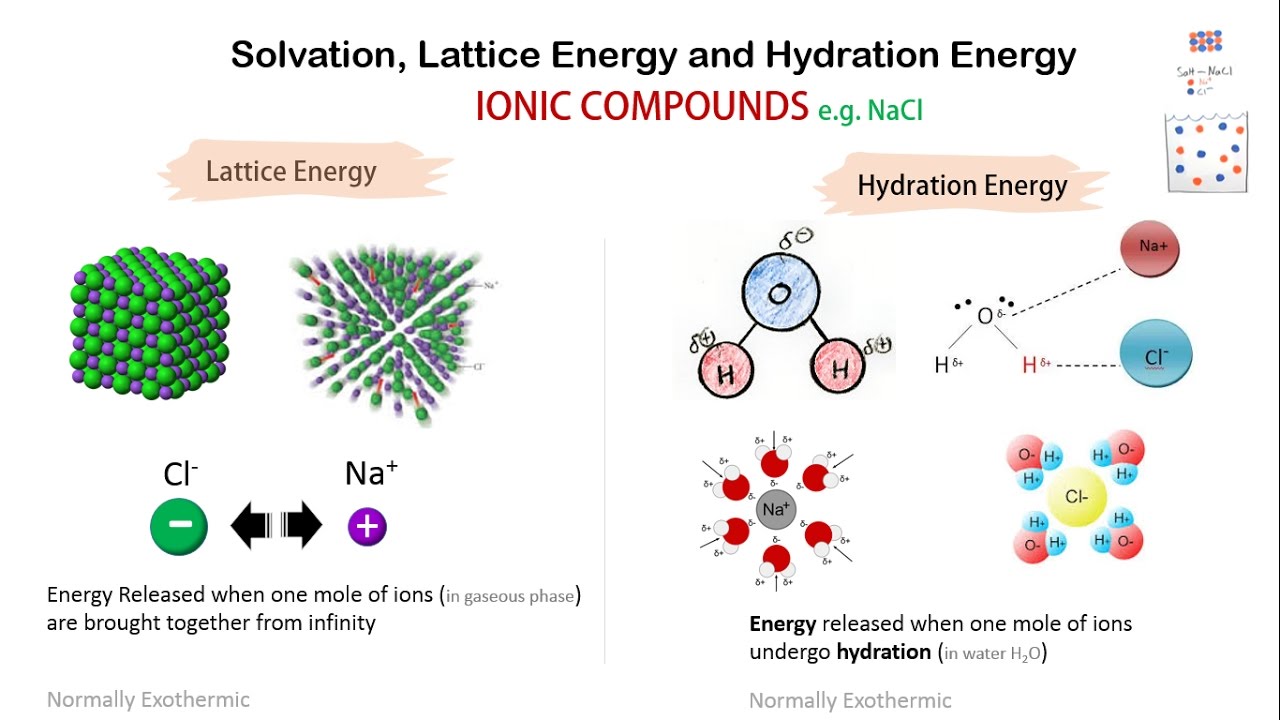
Reactivity (kinetics) and tendency of reaction outcome (thermodynamics) are independent. Potassium chloride ( KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine. That means that we will have to use theoretical values of their lattice enthalpies.Note that element electronegativity $\chi$ is a combined indicative quantity from covalent bond context, so its comparative values do not always apply in scenario analysis what happens or should be preferred. Let's look at this in terms of Born-Haber cycles of and contrast the enthalpy change of formation for the imaginary compounds MgCl and MgCl 3. Crystals cleave easily in three directions. It turns out that MgCl 2 is the formula of the compound which has the most negative enthalpy change of formation - in other words, it is the most stable one relative to the elements magnesium and chlorine. It adopts a face-centered cubic structure known as the B1 phase with a lattice constant of roughly 6.3. Thus lattice energy increases as the size. The question arises as to why, from an energetics point of view, magnesium chloride is MgCl 2 rather than MgCl or MgCl 3 (or any other formula you might like to choose). Lattice energy is directly proportional to the charge of the ions and inversely proportional to the size of the ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
